Monoclonal vs. polyclonal antibodies: How to choose
Antibodies, or immunoglobulins, are glycoproteins that play a major role in the immune response. They are produced by plasma cells, which are cells derived from the differentiation of B lymphocytes. Each antibody has a binding site, called “paratope”, which ensures the specific recognition of the target antigen. The part of the antigen recognized by the paratope is called “epitope”.
Antibodies are composed of two heavy chains and two light chains. They have a variable region responsible for antigen recognition. They also consist of a constant region (Fc portion), which plays a role in complement activation and in the elimination of immune complexes by cells expressing Fc receptors.
These glycoproteins, central to immunity, have become major tools in clinical practice, laboratory research, and therapy. In this context, distinguishing between monoclonal vs polyclonal antibodies is necessary to select the solution best suited to research needs.
This article explains what polyclonal and monoclonal antibodies are, then details their differences and their applications in research and medicine.
Monoclonal antibodies: Production and significance
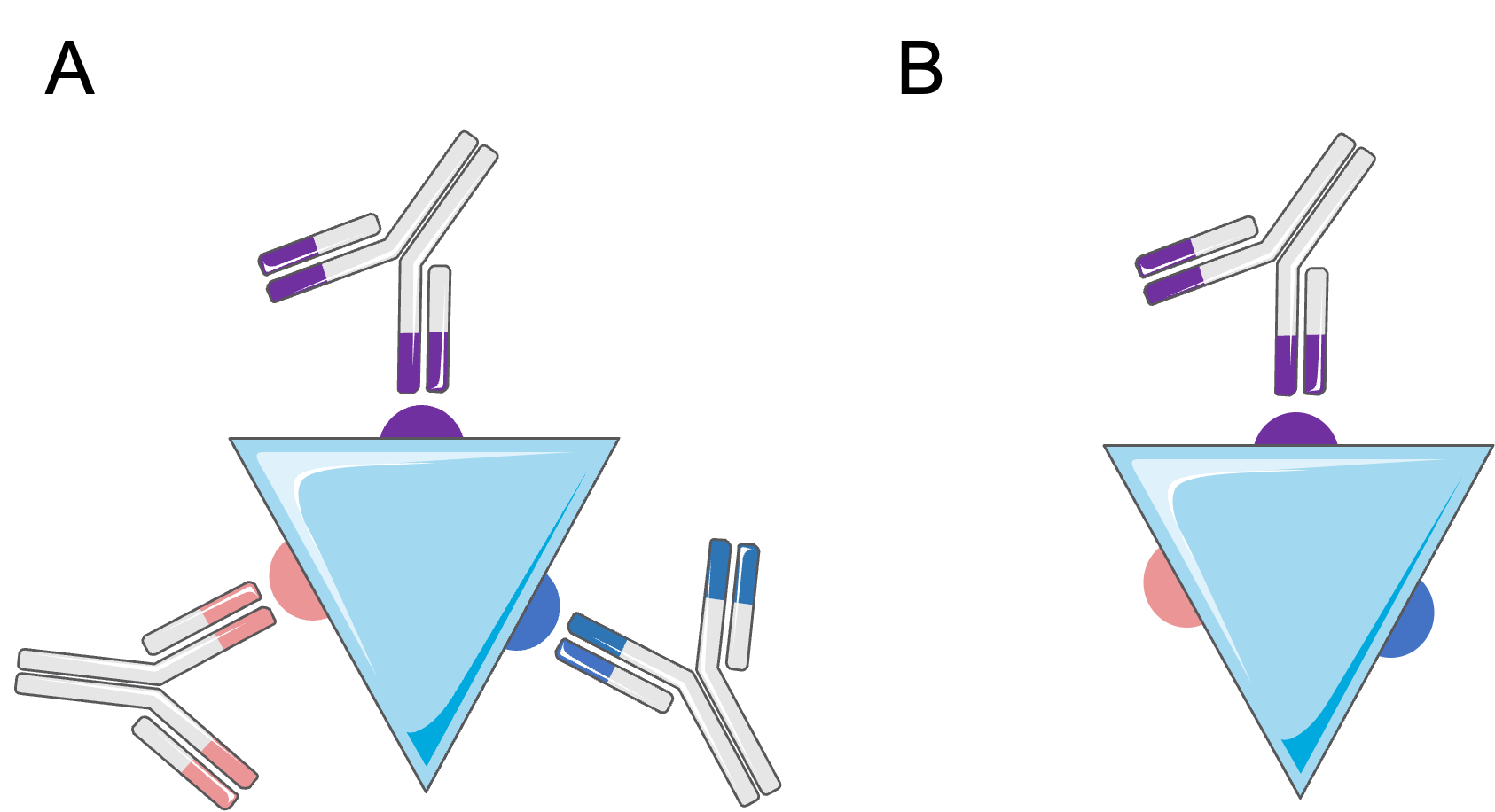
Monoclonal antibodies, produced by a single B-cell clone, are identical and directed against a single epitope of a given antigen. They are initially produced using hybridoma technology, which involves the fusion of immunized B cells with myeloma cells, resulting in an immortal cell line that secretes a single antibody.
The hybridoma technique provides a stable and renewable source of strictly identical (homogeneous) antibodies produced by a single cell clone. Each antibody recognizes a single specific epitope on the antigen, offering high specificity and affinity for the target. This technique was first developed by Georges Köhler and César Milstein (Köhler and Milstein, 1975), thereby revolutionizing the scope of application of these immunoglobulins.
Unlike polyclonal antibodies, which vary from batch to batch because they originate from multiple B-cell clones, monoclonal antibodies ensure high reproducibility and standardization of experimental results.
In research, monoclonal antibodies are preferred for the following reasons:
- specificity (targeted against a single epitope), for example to distinguish between two protein isoforms or phosphorylation states;
- batch-to-batch reproducibility, which is important for standardized quantitative assays (ELISA, Western blot, immunohistochemistry, etc.)
- purity and stability for approaches such as crystallography.
The monospecificity of monoclonal antibodies is useful when small structural changes need to be detected.
In medicine, most therapeutic antibodies are monoclonal antibodies. These biologics are used in the treatment of numerous diseases (oncology, rheumatology, immunology, etc.)
Monoclonal antibodies have been progressively humanized to limit immunogenicity and improve their half-life, which has enhanced their tolerability and clinical efficacy.
Polyclonal antibodies: Production and significance
Polyclonal antibodies are produced by multiple B-cell clones. They are directed against a single antigen but recognize several different epitopes.
Polyclonal antibodies are obtained by immunizing an animal (rabbit, goat, sheep, or chicken) with an antigen of interest. The serum, which contains all the immunoglobulins produced following the immune response, is then collected and purified to obtain antibodies specific to the antigen.
Polyclonal antibodies offer several advantages in research:
- sensitivity, through the recognition of multiple epitopes;
- robustness against changes in antigen structure (partial denaturation, post-translational modifications, etc.);
- avidity, by detecting antigens that are weakly expressed or present in low quantities.
Polyclonal antibodies are widely used in immunohistochemistry, immunofluorescence, and immunoprecipitation—methods in which the recognition of multiple epitopes enhances detection.
Historically, polyclonal antibodies have been central to serotherapy, with antitoxin or antivenom sera produced in animals. However, their therapeutic use is limited due to batch-to-batch variability and the need for numerous controls.
Monoclonal vs. polyclonal antibodies: The differences and how to choose
The comparison of monoclonal vs polyclonal antibodies is based on several parameters.
| Parameter | Monoclonal antibodies | Polyclonal antibodies |
|---|---|---|
| Origin | A single B-cell clone (hybridoma) | Multiple B-cell clones |
| Specificity | Recognizes a single epitope | Targets multiple epitopes of the same antigen |
| Homogeneity | Homogeneous, a single antibody molecule | Heterogeneous, a mixture of different antibodies |
| Production | Hybridomas, cell culture | Animal immunization, fast and affordable |
| Batch reproducibility | Excellent, unlimited supply | Varies depending on the animal and the batch |
| Sensitivity | Very good but depends on a single epitope | Very high due to multiple recognition |
| Tolerance to antigen variations | Low | Greater robustness (multiple targeted epitopes) |
| Typical uses in research | Specific assays, structural studies | Detection, immunohistochemistry (IHC), immunoprecipitation (IP), immunofluorescence (IF) |
| Uses in medicine | Targeted therapies, biopharmaceuticals, immunotherapy | Serotherapy, polyclonal immunoglobulins |
The choice between monoclonal vs polyclonal antibodies depends on the objective. If maximum specificity, excellent reproducibility, and a format suitable for the development of therapeutic drugs are required, monoclonal antibodies are the preferred choice. In general research, polyclonal antibodies offer robust detection, high sensitivity, and rapid, cost-effective production.
FAQ: Monoclonal vs. polyclonal antibodies
What is the difference between polyclonal and monoclonal antibodies?
Monoclonal antibodies derive from a single B-cell clone and recognize a single epitope. Polyclonal antibodies are a pool of antibodies produced by multiple clones that recognize multiple epitopes of the same antigen.
When should monoclonal antibodies be used in research?
Monoclonal antibodies are the best choice when high specificity, excellent batch-to-batch reproducibility, and highly specific recognition of an epitope (e.g., isoform, phosphorylation state, variant) are required.
What are the advantages of polyclonal antibodies in research?
Monoclonal antibodies are biopharmaceuticals able to neutralize cytokines, block receptors, or destroy specific cells (via ADCC or complement), and they have a major impact in oncology, rheumatology, gastroenterology, and neurology.
Why are monoclonal antibodies so widely used in medicine?
FcRn binding measures the interaction of antibodies with the neonatal Fc receptor at pH 6.0 and pH 7.4. These assays investigate the pH-dependent nature of the interaction which gives an indication of how efficiently antibodies are recycled instead of degraded, and to predict initial data about the half-life of the Drug i. Understanding FcRn binding is critical in therapeutic antibody development, as it guides strategies to extend circulation time and improve dosing regimens. Biacore™ SPR provides a robust, label-free way to study these interactions under physiologically relevant conditions.
What is a humanized monoclonal antibody?
CHumanized antibodies are recombinant antibodies whose structure has been modified to be predominantly or entirely human in order to reduce immunogenicity, prolong half-life, and improve clinical tolerance compared to the original murine antibodies.
Are polyclonal antibodies still used in therapy?
Yes, particularly in serotherapy (antitoxin or antivenom serums), in induction therapy for transplantation and in certain preparations of polyclonal human immunoglobulins derived from plasma, even though monoclonal antibodies now dominate the field of biopharmaceuticals.
References:
Köhler, G, and C Milstein. “Continuous cultures of fused cells secreting antibody of predefined specificity.” Nature vol. 256,5517 (1975): 495-7. doi:10.1038/256495a0
Neil S. Lipman, Lynn R. Jackson, Laura J. Trudel, Frances Weis-Garcia, Monoclonal Versus Polyclonal Antibodies: Distinguishing Characteristics, Applications, and Information Resources, ILAR Journal, Volume 46, Issue 3, 2005, Pages 258–268, https://doi.org/10.1093/ilar.46.3.258
Broutin, Watier, M., Hervé. Les Biomédicaments, 2ème Partie : Les Anticorps Thérapeutiques. Biologie Générale n°2 2016.